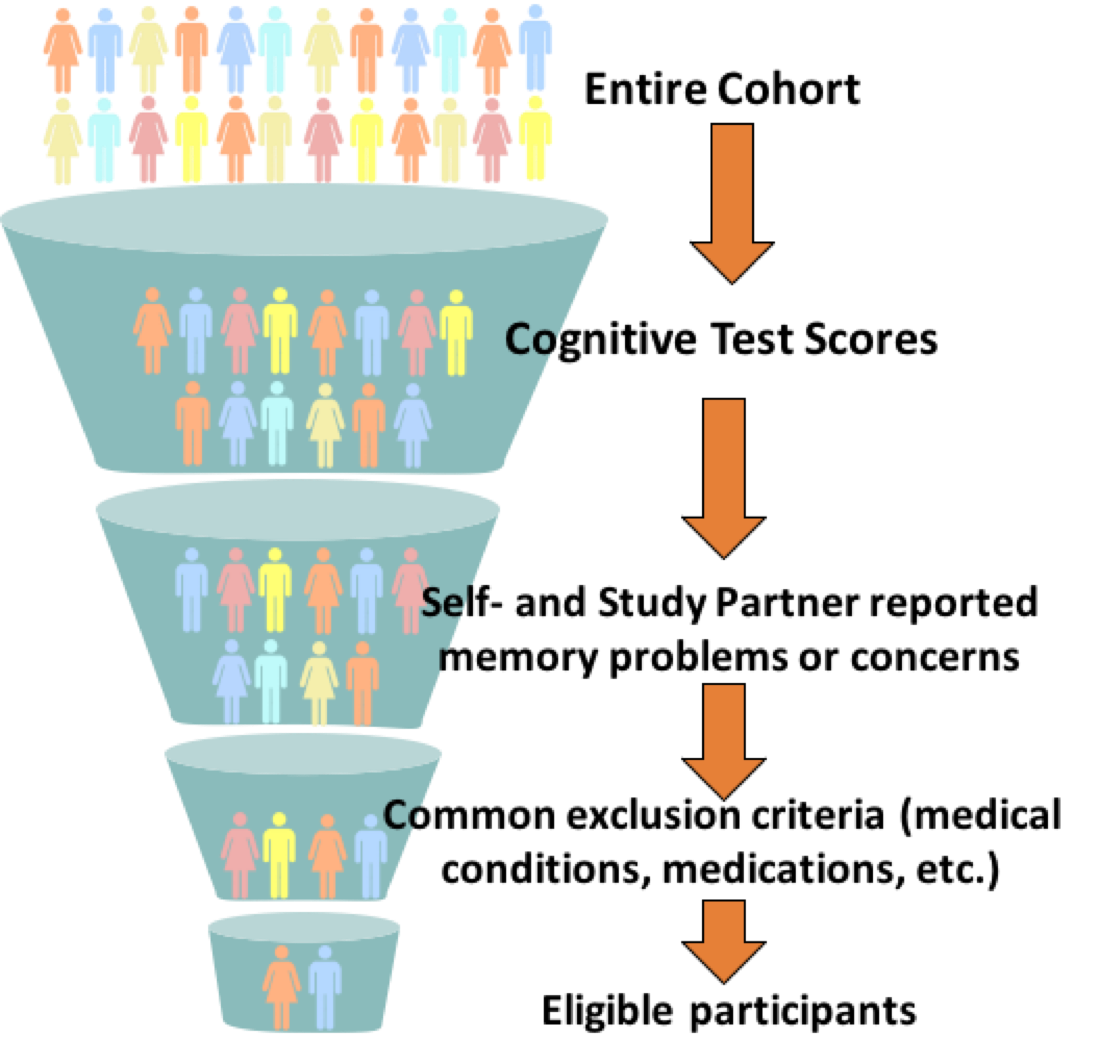
One of the goals of the Brain Health Registry is to assist with recruitment, assessment, and longitudinal monitoring of participants for neuroscience studies. We are eager to develop collaborations with other investigators who share the same goal of accelerating the development of improved diagnostic tests, effective treatments and preventative interventions for Alzheimer’s disease and other brain disorders.
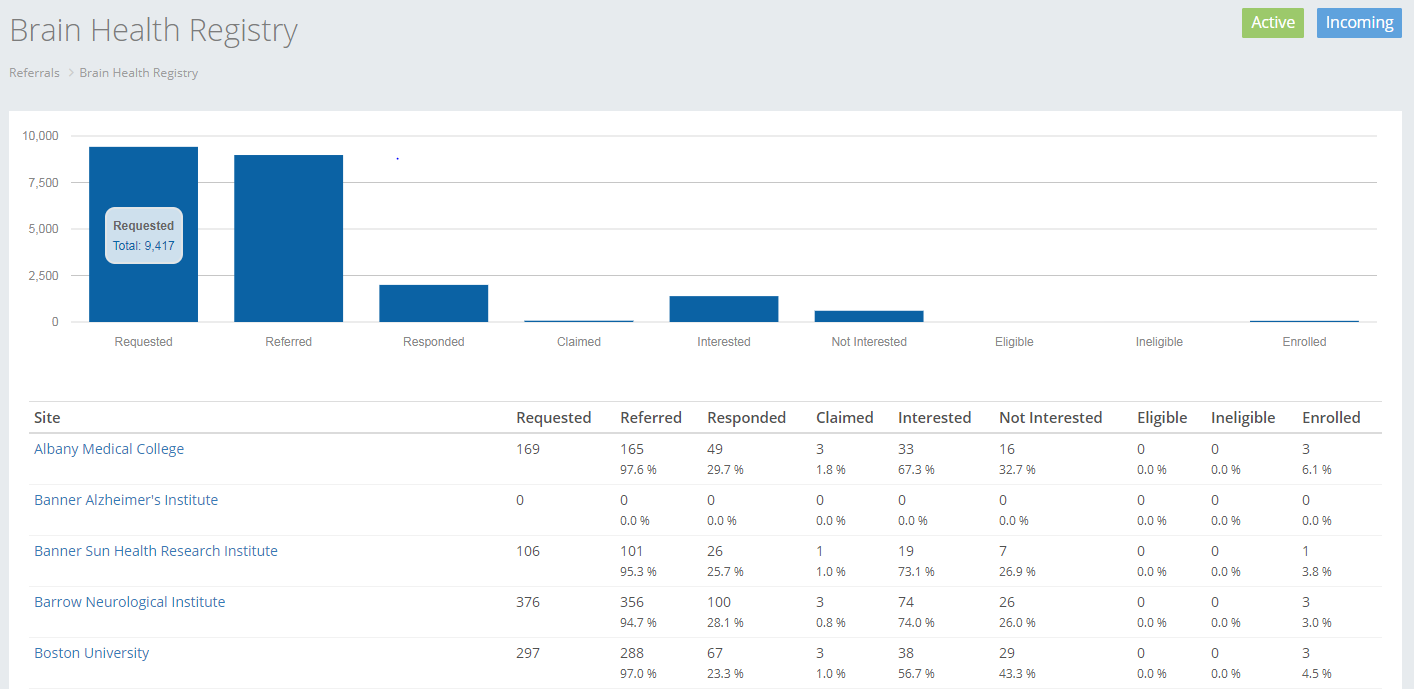
One of the ways that the Brain Health Registry can help facilitate clinical research is through a Comprehensive Referral Program. A Comprehensive Referral is the referral of existing BHR participants to other research studies.